Mass transport properties in quaternary systems at elevated pressures
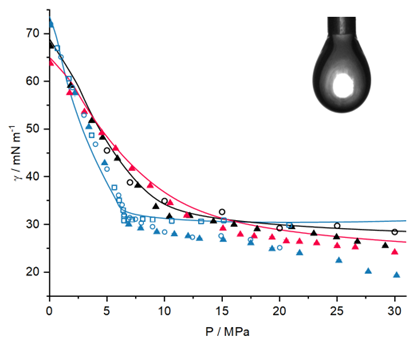
In the context of the energy transition, processes related to the storage and utilization of CO2 and H2, as well as fluid mixtures containing natural gas, are becoming increasingly important. These processes occur at elevated pressures, making it challenging to determine the respective phase equilibria and system properties such as interfacial tension and mass transfer. Experimental and theoretical investigations are conducted using the model systems “water – n-dodecane – n-butanol – CH4” and “water – n-dodecane – n-butanol – CO2,” which represent systems of high scientific interest, as it is known from the literature that two of the transferring compounds, n-butanol and CO2, are enriched at the interface. For the first time, two quaternary systems are systematically investigated in this project, i.e., all relevant thermodynamic properties are determined, including those of all binary and ternary subsystems. Furthermore, new experimental procedures are employed to systematically investigate mass transfer, which include the implementation of results from thermodynamic modeling that, in turn, provides a thermodynamically consistent approach based on PCP-SAFT. In combination with the density gradient theory (DGT), interfacial properties can be calculated. The ultimate goal of this project is to develop a thermodynamically consistent model for determining mass transfer in multiphase systems at elevated pressures. Furthermore, the database of fluid mixture properties is expanded, and new experimental methods are established.
Contact
M Sc. Rafael Villablanca
Department:
Petroleum Production Systems
Telephone: +49 5323 72 2560
Telefax: +49 5323 72 3146
E-Mail: rafael.villablanca@tu-clausthal.de
ADDRESS
Institute of Subsurface Energy Systems
Agricolastraße 10
Building C 21, Room 303
38678 Clausthal-Zellerfeld
